 Lymphocyte Development Group paper
Lymphocyte Development Group paper
Runx transcription factors help dictate the fate of T cellsas they become CD4+ or CD8+ T cells in the thymus, and Th1 orTh17 cells in the periphery. Now, according to Bruno et al.,Runx proteins also guide regulatory T (T reg) cell fate.
Runx proteins have been shown to bind to the canonical T regcell transcription factor Foxp3. Together, the two regulate(and primarily inhibit) the expression of target genes, suchas the Th17-promoting transcription factor Ror-γt. Now Brunoand colleagues reveal that Runx proteins also help induce andmaintain Foxp3 expression in mature CD4+ T cells.
Inducible T reg cells relied on Runx proteins to express Foxp3.Blocking the proteins reduced the number of these cells, andablating an indispensable subunit of Runx protein complexes,Cbfb, diminished Foxp3 expression in natural T reg cells. Runxproteins bound directly to the Foxp3 promoter in T reg cells,but not in naive CD4+ T cells because of locus inaccessibility.
Another group recently reported that Runx/Cbfb complexes controlthe expression of Foxp3 in natural T reg cells in vivo. WithoutCbfb, mice were susceptible to autoimmune disease. However,this study did not investigate a role for Runx proteins in Treg cell induction.
(Maxmen (2009) Runx: T reg cell keeper and creator. J. Exp. Med. doi:10.1084/jem.20611iti4)
This piece appears on the JEM website here
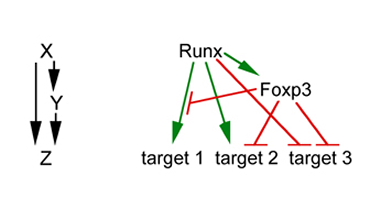
It was known from earlier studies that Foxp3 protein cooperates with Runx proteins in the regulation of target genes. The new finding that Foxp3 is itself regulated by Runx proteins suggests a regulatory circuit in which Runx proteins are both inducers and interaction partners of Foxp3. Genetic feed-forward loops involve two transcription factors: X regulates the expression of Y and both factors together regulate one or more downstream targets Z (left panel). In the case described by Bruno et al., X is a Runx family transcription factor and Y is Foxp3. Runx and Foxp3 interact at regulatory DNA sequences or independently of DNA to control the expression of several target genes (right panel). Feed-forward regulation is a recurring theme in the biology of Foxp3, which interacts not only with Runx but also other transcription factors including Rorγt and NFAT. This suggests that Foxp3 subverts T cell regulatory circuits to establish Treg cell-specific gene activation and repression programmes.
