Gut hormone therapies based on GLP-1 have transformed how we treat metabolic diseases. However, we still do not fully understand how these hormones signal the immune system to control inflammation. Our group investigates their action across the gut, brain and immune system, translating them into better therapies for chronic diseases.
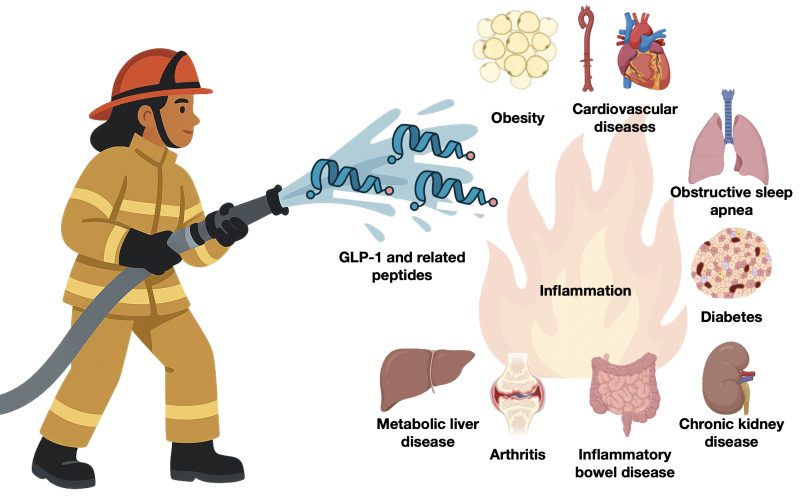
Chronic inflammation is a vicious cycle in many diseases: it damages vital organs and fuels more inflammation that spreads like a wildfire. While therapies based on the gut hormone GLP-1, which regulates blood sugar and appetite, have transformed how we treat metabolic disorders, their full potential to resolve inflammation remains an unanswered question.
Our group studies how gut hormones and related peptides broadly act as the body’s firefighters. By decoding how they change communication between organs, we can map exactly how they help resolve inflammation at its source. Our goal is to use these signals to protect the body, shifting from simply managing disease toward putting out the fire for good.
Our group studies how gut hormones regulate inflammation via the gut-brain-immune axis. We aim to decode the molecular crosstalk between these organs by pursuing the following three parallel directions.
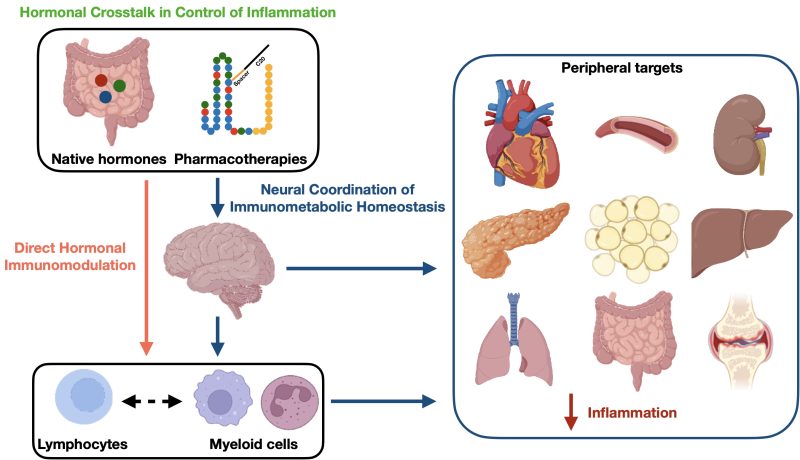
The clinical success of GLP-1 medicines has shown that gut hormones do much more than just control glucose or weight. By mapping how these signals function along the gut-brain-immune axis, we can translate our findings into two major goals for human health.
Wong, C.K., Drucker, D.J. (2025) Antiinflammatory actions of glucagon-like peptide-1–based therapies beyond metabolic benefits. Journal Of Clinical Investigation. doi:10.1172/JCI194751
Wong, C.K., Yusta, B., Tong, J.C.L., Broichhagen, J,. Hodson, D.J., Drucker, D.J. (2025). Reassessment of antibody-based detection of the murine T cell GLP-1 receptor. Cell Metabolism. doi:10.1016/j.cmet.2025.06.012
Wong, C.K., McLean, B.A., Baggio, L.L., Koehler, J.A., Hammoud, R., Rittig, N., Yabut, J.M., Seeley, R.J., Brown, T.J., Drucker, D.J. (2024). Central glucagon-like peptide 1 receptor activation inhibits Toll-like receptor agonist-induced inflammation. Cell Metabolism. doi:10.1016/j.cmet.2023.11.009
Wong, C.K., Yusta, B., Koehler, J.A., Baggio, L.L., McLean, B.A., Matthews, D., Seeley, R.J., Drucker, D.J. (2022). Divergent roles for the gut intraepithelial lymphocyte GLP-1R in control of metabolism, microbiota, and T cell-induced inflammation. Cell Metabolism. doi:10.1016/j.cmet.2022.08.003