By Helen Figueira
October 6, 2014
Time to read: 4 minutes
 Twenty-five years ago, the neuroscience community revealed the existence of neural stem cells, a life-long regenerative source of neurons and glia produced throughout life. The revelation has since generated great excitement within the field by opening up possibilities of developing therapeutic strategies for the treatment of brain injuries and neurodegenerative disorders. The concept of regenerative medicine, however, does rely on a thorough understanding of the basic principles of stem cell behaviour, which as yet are not fully understood. New research published today in Nature Cell Biology from the CSC’s Cell Interactions and Cancer group reveals novel insight into the signals and mechanisms involved in governing and regulating stem cell biology, which could pave the way for future regenerative therapies.
Twenty-five years ago, the neuroscience community revealed the existence of neural stem cells, a life-long regenerative source of neurons and glia produced throughout life. The revelation has since generated great excitement within the field by opening up possibilities of developing therapeutic strategies for the treatment of brain injuries and neurodegenerative disorders. The concept of regenerative medicine, however, does rely on a thorough understanding of the basic principles of stem cell behaviour, which as yet are not fully understood. New research published today in Nature Cell Biology from the CSC’s Cell Interactions and Cancer group reveals novel insight into the signals and mechanisms involved in governing and regulating stem cell biology, which could pave the way for future regenerative therapies.
Dr Simona Parrinello (Group Head, Cell Interactions and Cancer) and her team are interested in how the stem cell microenvironment – or niche – controls the fate of a stem cell. The vast majority of neural stem cells are in a quiescent state, waiting – throughout a lifetime – for their time to differentiate. Once they commit to differentiation, stem cells begin to divide and later generate neurons. Blood vessels in the niche have been known to secrete chemicals, or signals, which promote division and differentiation of committed stem cells. Yet, a recent study characterising the precise structure of the niche revealed that quiescent stem cells are found in direct contact with blood vessels, indicating that they should be exposed to these pro-differentiation signals, but are somehow resistant to them.
To investigate this apparent paradox, Simona and her team delved right into the niche’s vasculature and uncovered something new about the role of blood vessels in stem cell function. They discovered that at the sites of contact specific signals were being presented to the stem cells by the blood vessels, which was keeping the stem cells in a quiescent state despite being surrounded by cell differentiating chemicals.
They identified the signals as cell surface-bound proteins, known as ephrinB2 and jagged1, that are only functional when cells are in direct physical contact with one another. Simona’s group propose that when quiescent cells are then ready to differentiate, the contact is broken allowing them to respond to the differentiation promoting chemicals within the niche and journey towards neuron formation.
“This is the first time we have identified the blood vessels as a key niche component for maintaining a pool of undifferentiated stem cells throughout life to sustain neurogenesis (the birth of new neurons) long-term,” explains Simona. “One of the central questions of the stem cell field is how the niche can both maintain stem cells in the quiescent state, while at the same time promote stem cell differentiation along the correct linage.Our findings provide a mechanistic explanation of how the niche is able to direct such opposite stem cell behaviors,” she adds.
The niche has been proposed as an ideal target for regenerative therapies, as it is more ‘druggable’ than the stem cells themselves and should allow for safer, extrinsic modulation of stem cell function. This study has successfully identified two candidates for such ‘niche therapies’, which could be targeted to transiently boost neurogenesis for therapeutic purposes.
Publication: Ottone C, Krusche B, Whitby A, Clements M, Quadrato G, Pitulescu ME, Adams RH & Parrinello S: Direct cell–cell contact with the vascular niche maintains quiescent neural stem cells, Nature Cell Biology doi:10.1038/ncb3045, published online
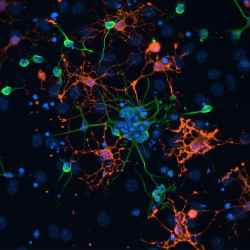
Image: Neural stem cells differentiated in vitro into neuroblasts (green) and oligodendrocytes (red).
This work was supported by the Medical Research Council, Imperial College London and the Royal Society. The Medical Research Council has been at the forefront of scientific discovery to improve human health. Founded in 1913 to tackle tuberculosis, the MRC now invests taxpayers’ money in some of the best medical research in the world across every area of health. Twenty-nine MRC-funded researchers have won Nobel prizes in a wide range of disciplines, and MRC scientists have been behind such diverse discoveries as vitamins, the structure of DNA and the link between smoking and cancer, as well as achievements such as pioneering the use of randomised controlled trials, the invention of MRI scanning, and the development of a group of antibodies used in the making of some of the most successful drugs ever developed. Today, MRC-funded scientists tackle some of the greatest health problems facing humanity in the 21st century, from the rising tide of chronic diseases associated with ageing to the threats posed by rapidly mutating micro-organisms. www.mrc.ac.uk