By Sophie Arthur
January 14, 2020
Time to read: 4 minutes
What comes to mind when you hear ‘schizophrenia’? Hallucinations and delusions? Cognitive impairment? Feeling withdrawn from everyone around you? Apathy? With all the above being symptoms of this mental disorder, and with 1 in every 100 people affected, each individual’s life becomes a relentless battle, struggling with habitual activities, social interaction, and general wellbeing and self-care.
Current treatments help to reduce the severity of symptoms and allow patients to maintain a more functional daily life. However, antipsychotic medications, which are typically required lifelong, are not guaranteed to work for all schizophrenia patients, with only about 4 in 5 people benefitting usually.
Great efforts have gone into exploring the biology underlying the condition to try and combat this issue, with the latest research from the MRC LMS focusing on synapses. Synapses are the junctions between neurons. Neurons communicate across the synapse via neurotransmitters; chemicals that transmit the nerve signal. Researchers investigated the relationship between synaptic density and schizophrenia, questioning whether a reduction in these brain connections are present in schizophrenia, and whether they result from treatment with antipsychotic drugs. The study was inspired by findings from previous post-mortem studies that demonstrated synaptic reductions in the brains of schizophrenia patients compared to healthy controls.
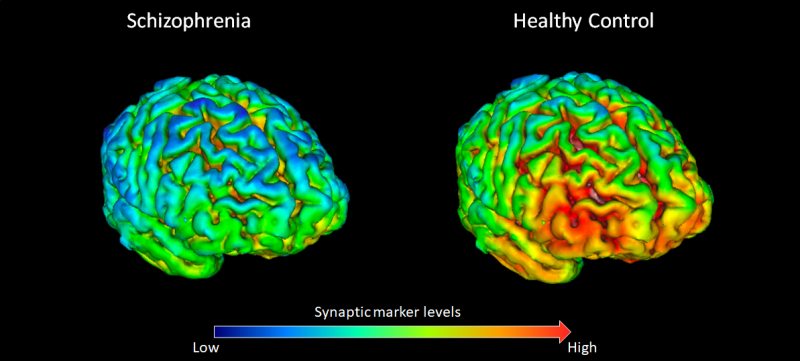
Measuring synaptic protein levels in living patients was not possible before the development of a new PET (Positron Emission Tomography) radioactive tracer that binds to a specific protein associated with synapses. Being “one of the first centres in the world” to have access to this, Professor Oliver Howes, Head of the Psychiatric Imaging Group at the MRC LMS, and his team grasped this opportunity to investigate synaptic loss in schizophrenic brains through the synaptic marker protein SV2A, which is found on the nerve terminal preceding the synapse and is involved in regulating neurotransmitter release.
The team found lower levels of SV2A in the brains of schizophrenia patients, indicating a reduction of synapses. Certain areas exhibited significantly lower levels, such as the frontal cortex which coordinates use, recall and processing of memory, planning complex cognitive behaviour and personality expression. These findings were supported by the understanding that these areas are associated with some of the major cognitive impairments seen in schizophrenia, thus providing a potential clue that synaptic loss may underlie these problems.
But in order to test whether the loss was a result of the illness or in fact due to treatment, a preclinical study was conducted in rat models investigating the effect of antipsychotic drugs on the levels of SV2A. They found that the antipsychotics did not contribute to the synaptic loss seen in schizophrenic patients, thereby reinforcing their overall findings.
So, what does this mean for the development of schizophrenia treatment? If synaptic loss is an underlying key contributor in the disease, further research into the mechanisms could lead to exploring ways to prevent this loss. Moreover, since microglia, known as the immune cells of the brain, play a role in mediating synaptic pruning, there is also potential to target microglia and see if it could introduce a way of preventing excess synaptic loss; an investigation that the team have already begun.
“The human brain, with its approximately 100 trillion synapses, is an extraordinarily complex organ,” says Dr Ellis Chika Onwordi of the Psychiatric Imaging Group and first author of this study. “Having the means to characterise the distribution of these synapses in the living brain, and to find differences in synaptic distribution between patients with schizophrenia and healthy controls, represents a significant advance in our ability to study the neurobiology of schizophrenia.”
“Synaptic density marker SV2A is reduced in schizophrenia patients and unaffected by antipsychotics in rats” was published in Nature Communications on 14 January. Read the full article here.
Written by Emily Jin