By Sophie Arthur
September 25, 2019
Time to read: 4 minutes
Idiopathic pulmonary fibrosis (IPF) is a type of lung disease that results in scarring of the lungs, also known as fibrosis. Over time, the fibrosis gets increasingly worse so that it becomes hard for those with the condition to simply take in a deep breath to get the oxygen they need. However, it is not clear what the cause of this condition is, and while there are two treatments that can help slow the rate at which IPF progresses, there is currently no treatment that can stop or reverse the scarring of the lung. However, recent research may be paving the way towards a new approach for the treatment for IPF.
Research published in the journal Science Translational Medicine on 25 September by members of the Cardiovascular Disease Mechanisms group here at the MRC LMS in collaboration with Duke-NUS Medical School, National Heart Centre Singapore & National Heart and Lung Institute, Imperial College London, showed that blocking a protein called interleukin-11 (IL-11) using therapeutic antibodies can reverse the fibrosis in the lung.
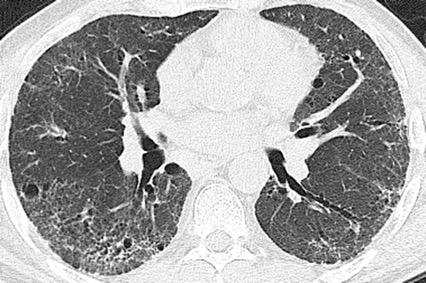
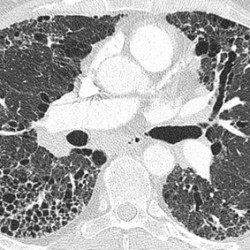
CT scan from an individual with IPF at diagnosis (top) and 18 months later (bottom) after the disease as progressed a lot
Hidden in plain sight
In 2013, Toby Maher; one of the authors on this paper, working with his colleague Elizabeth Renzoni, independently found that the expression of IL-11 was highly upregulated in this condition. In fact, it was the most highly expressed of all genes in lung fibroblasts, a specific type of structural cell which is capable of producing scar tissue, obtained from the lungs of patients with IPF. However, back then, the function of IL-11 was unclear, and there was no mention of IL-11 in relation to this disease previously. It wasn’t until 2017 that the true biology of IL-11 was discovered. Only then did it become clear how important upregulation of IL-11 might be in IPF, as IL-11 causes lung fibroblasts to become highly active and to destroy the lung causing it to scar and not function.
In this study, the teams investigated whether drugs called therapeutic antibodies could reverse the process of lung fibrosis, as IL-11 was found to be crucial to the progression of IPF. Using mice designed to mimic patients suffering from the lung disease, antibodies designed to bind to IL-11 and neutralise its activity were administered. This treatment not only diminished the damage of the lungs in these mice, but reversed the fibrosis too, highlighting the potential of IL-11 as a possible target for the treatment of IPF. This is in addition to its potential in the liver, which was reported by the same group earlier this year to cause liver scaring, which could also be reversed with antibody therapy.
Stuart Cook, Head of the Cardiovascular Disease Mechanism group at the MRC LMS and senior author of this study discussed the exciting next steps for this research:
“We found that blocking the IL-11 protein with antibodies could reverse fibrosis in a mouse model of human lung disease. This is a remarkable finding as reversing fibrosis is tough to do. We believe this holds promise for treating fibrotic lung diseases, like IPF, in patients. We are aiming to test our antibodies in human safety trials by the end of 2020, and then start clinical trials with patients in 2021.”
Toby Maher, Professor of Interstitial Lung disease and British Lung Foundation Chair in Respiratory Research at NHLI explained the importance of this research for patients:
“Idiopathic respiratory failure is a terrible disease with an untreated life expectancy of 3 years. Although we have treatments to slow disease progression, we desperately need new therapies to genuinely transform outcomes for people with IPF. This exciting research highlights the importance of IL-11 in driving the development of fibrosis and gives hope for a new treatment approach to halt and maybe even reverse the devastating lung scarring of IPF.”
“Interleukin 11 is a therapeutic target in idiopathic pulmonary fibrosis” was published in Science Translational Medicine on 25 September. Read the full article here.