By Sophie Arthur
November 21, 2019
Time to read: 5 minutes
Eren Akademir is a PhD student in our Reprogramming and Chromatin group. Earlier this year, Eren entered the MRC Max Perutz Science Writing Award 2019 with this piece about his research exploring the intricate control of the development of sex cells.

It’s time for us to have the talk. You are at that age now. You see, when two people love each other very much, they… well what’s a sensitive way to put this? You’ve probably had the conversation already with your parents and I don’t want to make this as awkward as it was then, so I’ll be frank. I want to talk to you about sex. Now, I know what you’re thinking, “Oh dear writer, I already know lots about sex, thank you.” However, I’ll go a little bit deeper than the ‘birds and the bees’, to the mouse and my research about the wondrous development of sex cells.
We create new life through the fusion of gametes – the male’s sperm fertilising the egg from the female. Gametes are the part of you that is ‘immortal’, carrying a fraction of your genetic information from generation to generation.
But unfortunately, not for everyone. Significant numbers of men and women are faced with the cruel reality that they are unable to reproduce. According to NHS statistics, around 1 in 7 couples are affected by infertility problems.
Some find a solution through in vitro fertilisation (IVF), which involves mixing gametes from parents in the lab before implanting the resulting embryo back into the mother. IVF is not without its drawbacks though, with significant expense and low success rates for older women especially. The Human Fertilisation and Embryology Authority have recently criticised IVF clinics for “trading on hope.”
How do we help these people desperate to have their own child?
I am investigating aspects of these problems in mouse models by examining the remarkable journey cells take to form gametes during embryo development.
Let’s just take a second to comprehend how truly extraordinary gametes are. They are the only cells with the potential to produce a zygote – the founding master cell that divides to make up a whole organism. Just think – your organs, nervous system and of course your gametes too – all from that one cell. How is this possible? Epigenetics.
Our complete DNA genome is often compared to a manual but I prefer to see it as the voluminous manuscript for a novel like ‘In Search of Lost Time’. Although DNA isn’t written in extravagant French prose, it still uses 4 letters for its coded language: A, C, T and G. Nevertheless, like Proust’s epic, the text of DNA is extremely long, arranged into readable sentences and paragraphs of genes, and compacted into multiple volumes of chromosomes.
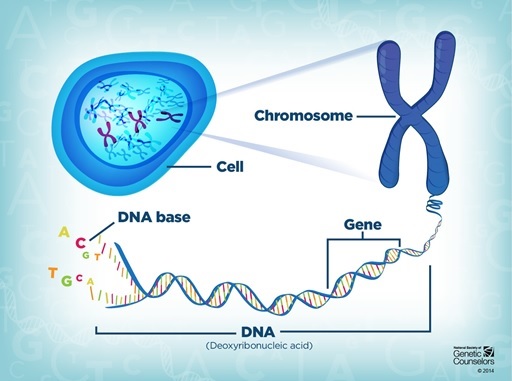
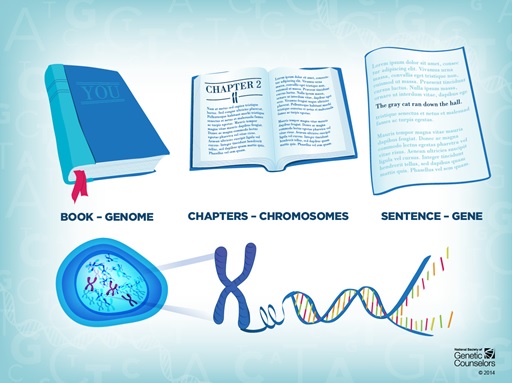
Every cell in your body has the same copy of the genome but each interprets it differently. This is where epigenetics comes in to add another layer, providing tools to annotate on the DNA manuscript.
Sentences of genes could be highlighted for activation, or conversely, crossed out to be switched off using a chemical tagging system called methylation. In fact, these tags often glue whole chapters together, making them inaccessible; never to be read. These superficial modifications to the manuscript restructure the whole novel, changing the story of the cell’s fate.
I research the intriguing story of the early sex cells called ‘primordial germ cells’ (PGCs). To become gametes, PGCs must undergo a unique epigenetic phenomenon called ‘reprogramming’. They deploy erasers that work across the whole genome, wiping away notes of methylation pencilled on the DNA previously.
This genome-wide erasure in PGCs must be tightly controlled, otherwise parts of the script necessary to make a gamete later are left unreadable. Disturbance of this process could be responsible for many cases of infertility and poor IVF success rates.
What could influence the methylation erasure?
Like many diseases, metabolism could be a critical factor. But to explore this further, I need to find a way to measure the methylation tags across the genome.
I do this by using powerful machines called mass spectrometers, which overload the DNA with heaps of energy, fragmenting them into their single letter form. We can then calculate the proportion of tagged and non-tagged letters based on the mass of the shattered parts.
My experiments have looked at how the “ink” for tags come from a compound called methionine, an essential component of our diet derived from protein. Methionine depletion reduces the amount of tags throughout the DNA. However, PGCs can potentially make the ink from other sources in the environment too. It’s up to me to decipher this supply network.
As our understanding of epigenetic reprogramming in the mouse continues to improve, so will our ability to make human gametes fertile in the lab. Metabolism may just be a key piece in the jigsaw to making babies for these couples struggling with infertility.
So, when the time comes to sit your child down and tell them about the birds and the bees, don’t forget the mice too!