“What is the link between the processes that drive cell metabolism and disease?”
The biological fuel for cells is adenosine triphosphate (ATP) – a molecule that when broken down, releases energy. ATP powers a wide range of processes in living cells and is essential for cell survival.
When a cell is under stress and ATP levels begin to fall, an enzyme called AMP-activated protein kinase (AMPK) kicks into gear.
AMPK is often referred to as the “fuel gauge” of the cell, as it increases generation of ATP whilst decreasing its utilisation to keep ATP levels constant. This process is controlled by a number of signalling pathways, and if mis-regulated, can cause metabolic diseases, including cancer.
Through our research, we hope to find out more about the regulation of AMPK and the role it plays in the development of human disease, and how it may also help treat disorders and health conditions.
We use a number of approaches including biochemical studies, molecular biology and physiology. We developed a transgenic mouse model that has a mutation in AMPK leading to an increase in AMPK activity. Using this model, we can study how AMPK activation in different disease settings, such as obesity and cancer, impacts on metabolism and disease progression. As an alternative approach, we can use newly developed small molecule AMPK activators to determine how they alter metabolism and whether they can protect against disease progression. These studies provide valuable insight into the therapeutic potential of activating AMPK.
We hope that our research will pave the way for the development of treatments for a range of diseases, including certain cancers and metabolic diseases such as obesity and fatty liver disease.
“Our research is aimed at understanding the regulation of energy metabolism”
AMP-activated protein kinase (AMPK) is activated in response to a fall in the intracellular concentration of ATP, and has been dubbed the cell’s fuel gauge. AMPK phosphorylates a wide range of downstream targets involved in diverse pathways. Broadly speaking, activation of AMPK leads to an increase in catabolic pathways and a reduction in anabolic pathways, effects that are predicted to be beneficial in metabolic diseases. As a consequence, AMPK has attracted significant attention as a potential therapeutic target for drugs aimed at treating metabolic diseases, including obesity and type 2 diabetes.
Phosphorylation and activation of AMPK is catalysed by either LKB1 or Ca2+/calmodulin dependent protein kinase kinase 2 (CAMKK2). Inactivating mutations within LKB1 lead to a rare, dominantly inherited cancer predisposition in humans, termed Peutz-Jeghers Syndrome, whereas CAMKK2 expression is increased in human prostate cancer. These findings suggest that AMPK may play a role in human cancers. Our current focus is to understand the regulation of AMPK using structure/function analyses and to explore its physiological role using a recently developed transgenic mouse model expressing a gain-of-function AMPK mutant.
An exciting new development is our finding that AMPK activation protects against diet-induced obesity by increasing thermogenesis in white adipose tissue (WAT). Importantly, the mechanism for increased thermogenesis does not involve UCP1, but appears to utilise a calcium cycling mechanism. A key challenge for the future is to identify the cells responsible for the non-UCP1mediated thermogenesis in WAT and to characterise the molecular mechanisms controlling their growth and differentiation.
BBSRC (fund Alice Pollard) and BHF (fund Sophie Newton)
We study the role of the enzyme AMPK (AMP-activated protein kinase) in regulating metabolism. AMPK acts as the cell’s energy sensor, monitoring the level of ATP and coordinating metabolic pathways to ensure ATP levels are sustained. Dysregulation of energy metabolism occurs in a wide range of human diseases, including cancer, obesity and neurodegeneration.
Our work aims to provide fundamental information on how AMPK regulates metabolic pathways, and how this can be harnessed in treating or preventing metabolic diseases. This knowledge is essential for the development of new and improved strategies for combatting these conditions. We use a combination of approaches and techniques, ranging from structural studies, cell-based assays and in vivo mouse models. We collaborate with Pharma to explore the therapeutic utility of small molecule modulators of the AMPK pathway.



Widjaja, A. A., Lim, W., Viswanathan, S., Chothani, S., Corden, B., Dasan, C. M., Goh, J. W. T., Lim, R., Singh, B. K., Tan, J., Pua, C. J., Lim, S. Y., Adami, E., Schafer, S., George, B. L., Sweeney, M., Xie, C., Tripathi, M., Sims, N. A., Hübner, N., Petretto, E., Withers, D. J., Ho, L., Gil, J., Carling, D., Cook, S. A. (2024). Inhibition of IL-11 signalling extends mammalian healthspan and lifespan. Nature, 632(8023), 157–165. https://doi.org/10.1038/s41586-024-07701-9
Penfold, L., Woods, A., Pollard, A.E., Arizanova, J., Pascual-Navarro, E., Muckett, P.J., Dore, M.H., Montoya, A., Whilding, C., Fets, L., Mokochinski, J., Constantin, T.A., Varela-Carver, A., Leach, D.A., Bevan, C.L., Nikitin, A.Y., Hall, Z., Carling, D. (2023). AMPK activation protects against prostate cancer by inducing a catabolic cellular state. Cell Reports, 42 (4), art. no. 112396.
Hope, D.C.D., Hinds, C.E., Lopes, T., Vincent, M.L., Shrewsbury, J.V., Yu, A.T.C., Davies, I., Scott, R., Jones, B., Murphy, K.G., Minnion, J.S., Sardini, A., Carling, D., Lutz, T.A., Bloom, S.R., Tan, T.M.M., Owen, B.M. Hypoaminoacidemia underpins glucagon-mediated energy expenditure and weight loss. (2022). Cell Reports Medicine, 3 (11), art. no. 100810.
Widjaja, A.A., Viswanathan, S., Wei Ting, J.G., Tan, J., Shekeran, S.G., Carling, D., Lim, W.-W., Cook, S.A. IL11 stimulates ERK/P90RSK to inhibit LKB1/AMPK and activate mTOR initiating a mesenchymal program in stromal, epithelial, and cancer cells. (2022). iScience, 25 (8), art. no. 104806.
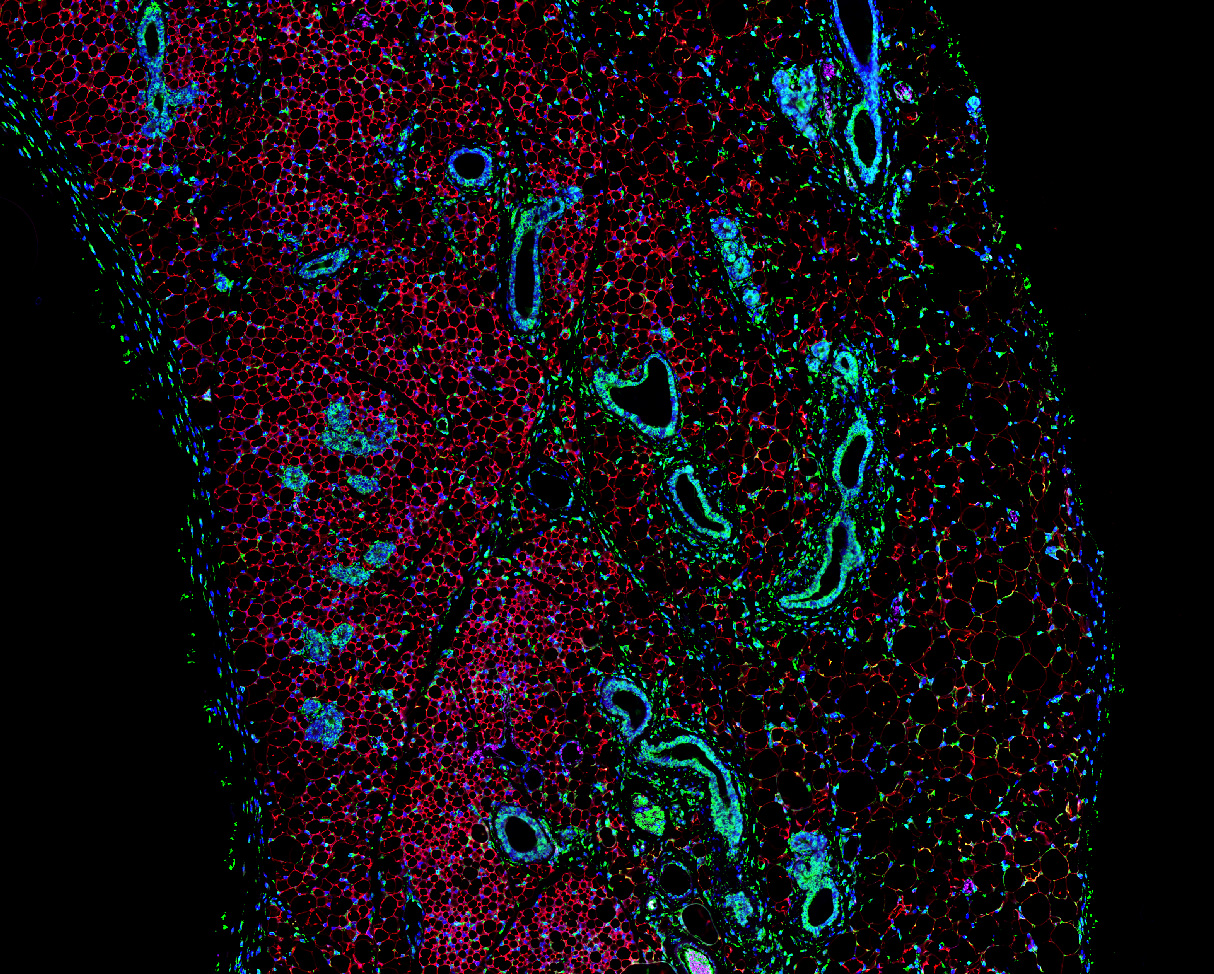
Wilson, L., Pollard, A.E., Penfold, L., Muckett, P.J., Whilding, C., Bohlooly, M.Y., Wilson, P., Carling, D. (2021). Chronic activation of AMP-activated protein kinase leads to early-onset polycystic kidney phenotype. Clinical Science, 135 (20), pp. 2393-2408.
Pollard AE, Martins L, Muckett PJ, Khadayate S, Bornot A, Clausen M, Admyre T, Bjursell M, Fiadeiro R, Wilson L, Whilding C, Kotiadis VN, Duchen MR, Sutton D, Penfold L, Sardini A, Bohlooly-Y M, Smith DM, Read JA, Snowden MA, Woods A and Carling D. (2019). AMPK activation protects against diet induced obesity through Ucp1-independent thermogenesis in subcutaneous white adipose tissue. Nature Metabolism, 1, 340-349.